Tag: Swallowing Assessment
-

BOLUS Framework
BOLUS Framework Quick Guide A Practical Dysphagia Reference for SLPs The BOLUS framework helps clinicians think beyond aspiration alone when evaluating the risk of an adverse event in patients with dysphagia. Aspiration does not automatically lead to aspiration pneumonia or pulmonary complications. Risk is influenced by the aspirated material, the patient’s host defenses, overall health,…
Written by
·
-

LSVT LOUD and Swallowing: How a Voice Treatment Helps Dysphagia
LSVT LOUD, originally for Parkinson’s voice issues, may also benefit swallowing due to shared anatomical structures. Research indicates it can enhance vocal fold closure, improve cough strength, and optimize neuromuscular control, leading to better swallowing efficiency. However, further studies are needed to establish its efficacy for dysphagia universally.
Written by
·
-

The Free Water Protocol: Why Sometimes All a Patient Wants Is Just… Water
The Free Water Protocol allows certain patients, particularly those on thickened liquids or NPO status, to safely consume regular water between meals. Emphasizing hydration and patient dignity, it incorporates oral care and eligibility criteria. Research shows it enhances hydration and patient satisfaction, countering dehydration and fostering a sense of normalcy in care.
Written by
·
-

How to Prevent Dehydration in Dysphagia: Evidence-Based Hydration Tools for SLPs
Discover safe, research-supported hydration techniques for dysphagia—ice chips, free water, moisture-rich foods, and oral care to reduce dehydration risk.
Written by
·
-

Flexible Endoscopic Evaluation of Swallowing (FEES) One Gold Standard Assessment
FEES (Flexible Endoscopic Evaluation of Swallowing) is an assessment procedure gaining recognition alongside the Modified Barium Swallow Study (MBSS). It allows real-time visualization of swallowing mechanics without barium, offering insights into aspiration and residue. Myths surrounding FEES, such as pain and safety, are debunked, reinforcing its value in dysphagia diagnosis.
Written by
·
-

30 Facts About Dysphagia to Raise Awareness and Improve Patient Care
June is Dysphagia Awareness Month, highlighting the condition affecting millions. Dysphagia, or difficulty swallowing, can stem from various underlying issues and leads to serious health risks. Speech-Language Pathologists play a vital role in diagnosis and treatment. Raising awareness about dysphagia improves outcomes for patients, caregivers, and healthcare providers alike.
Written by
·
-

Math and Dysphagia
The blog post by George Barnes and Doreen Benson emphasizes the importance of incorporating statistics into speech-language pathology (SLP) practices, specifically for treating dysphagia. It discusses common cognitive biases affecting clinical decision-making and stresses the need for a more analytical approach in evaluating patient risks and benefits, promoting better outcomes in care.
Written by
·
-
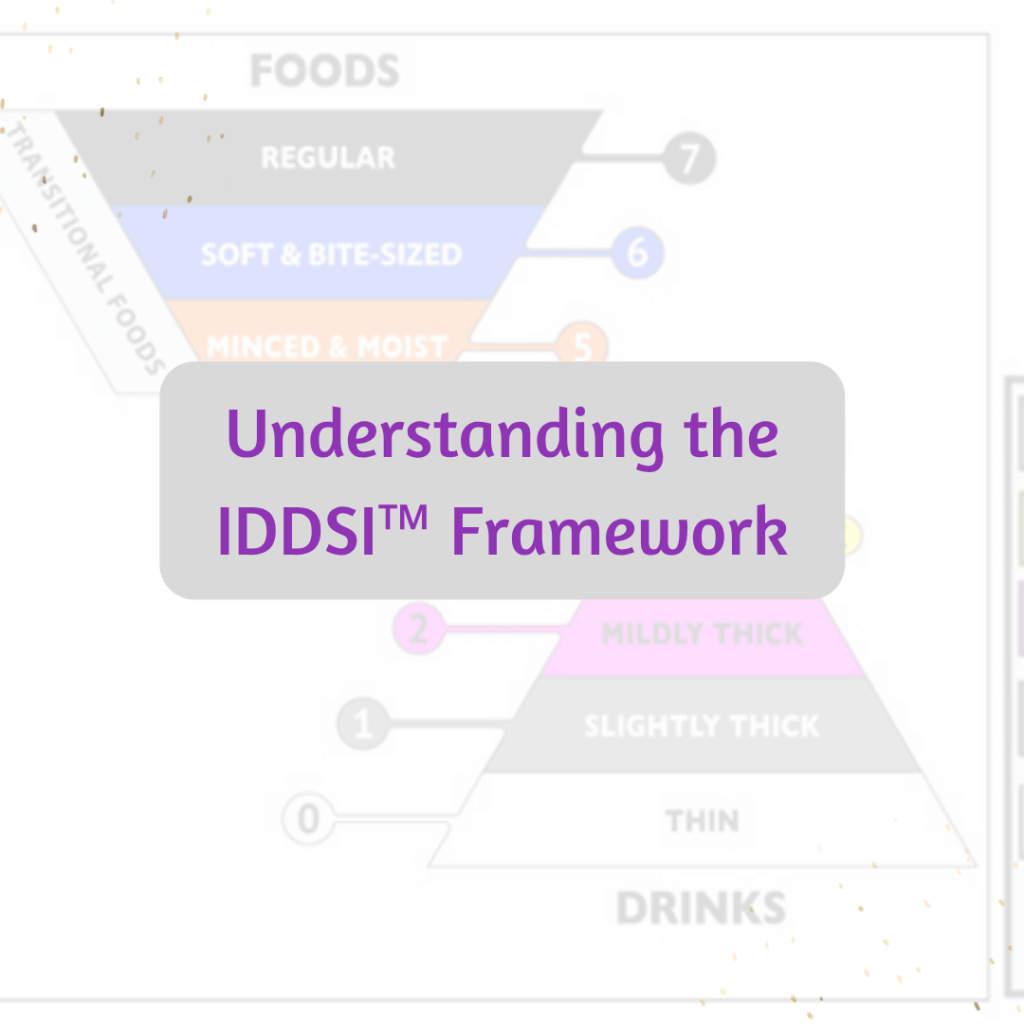
Understanding the IDDSI™ Framework
The International Dysphagia Diet Standardisation Initiative (IDDSI™) provides a unified system for food and drink consistencies, enhancing patient safety. It replaces the National Dysphagia Diet, offering clear levels for drinks and foods, minimizing risks. Resources such as testing cards and guides support implementation and training in diverse healthcare settings.
Written by
·
-

How Cognition Influences Swallowing Safety
Cognition significantly influences swallowing abilities, with cognitive decline leading to increased risks of aspiration and malnutrition. A clinical case illustrated that normalizing sodium levels improved swallowing function. Studies show that cognitive assessments predict aspiration risks, highlighting the importance of screening patients’ orientation and command-following before dysphagia evaluations to ensure safety in oral intake.
Written by
·
